Important Safety Information
Do not use VEREGEN® (sinecatechins) Ointment, 15% if you are allergic to any ingredient in this product. Do not use VEREGEN® Ointment, 15% for warts in the vagina, cervix, or inside the anus. Avoid contact with your eyes, nostrils and mouth while ointment is on your finger(s).
Avoid use of VEREGEN® on open wounds. Do not expose skin that has been treated with VEREGEN® to the sunlight, sunlamps or tanning beds. Tell your doctor if you are using any other type of skin product on the area to be treated. Avoid sexual contact (genital, anal or oral) when VEREGEN® Ointment, 15% is on your genital or perianal skin. If you do choose to have sexual contact, you must wash off the ointment carefully before having protected sexual contact as the ointment may weaken condoms and vaginal diaphragms.
Be sure to tell the doctor if you have a weak immune system, if you are pregnant or nursing a baby, or if you have used VEREGEN® before. Avoid using this product in patients younger than 18 years of age or for longer than 16 weeks. If your warts do not go away or come back after treatment contact your doctor.
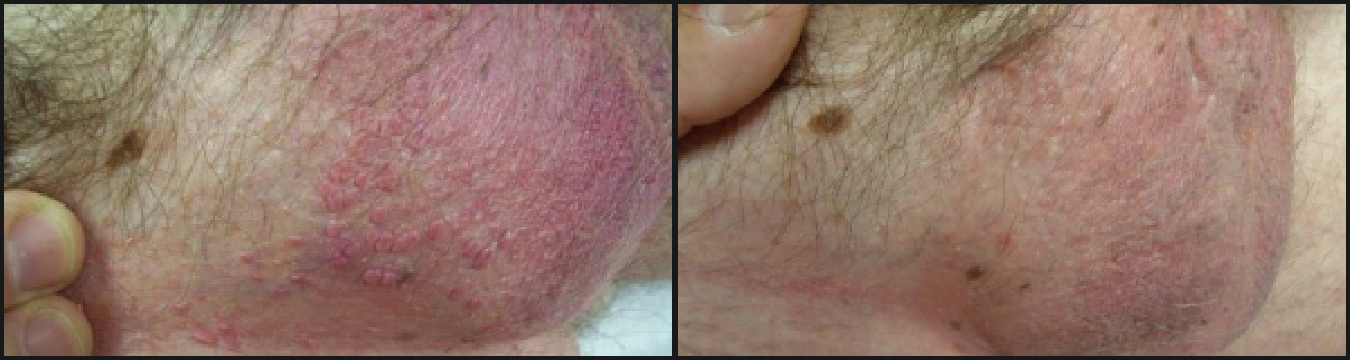
The most common side effects with VEREGEN® Ointment, 15% are local skin and application site reactions including (incidence ≥20%): redness, itching, burning, pain, sores, swelling, hard spots, and rash with blisters.
For more information, consult your healthcare professional.
Please see full Prescribing Information for VEREGEN®.
You are encouraged to report negative side effects of prescription drugs to the Food and Drug Administration (FDA). Please visit www.fda.gov/medwatch or call 1-800-FDA-1088.
Indication
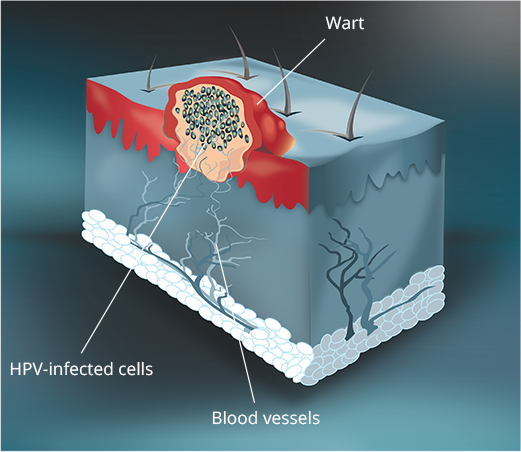
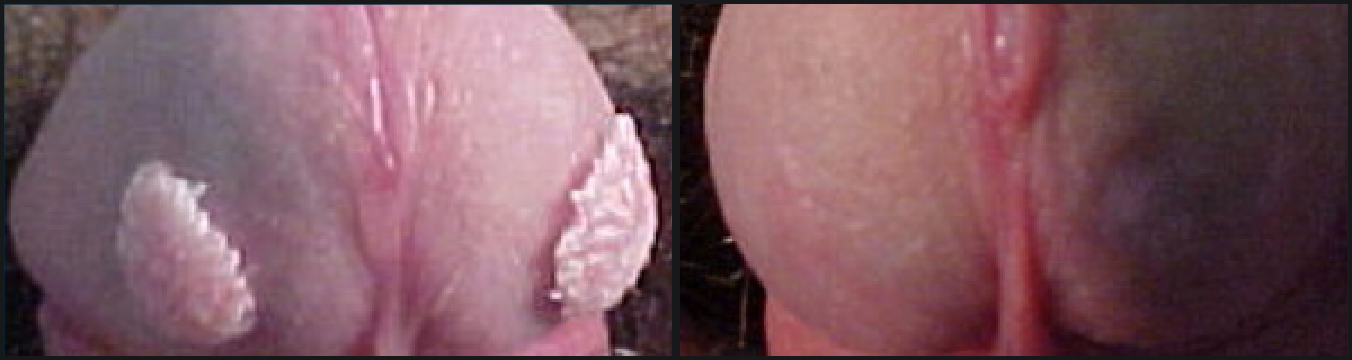
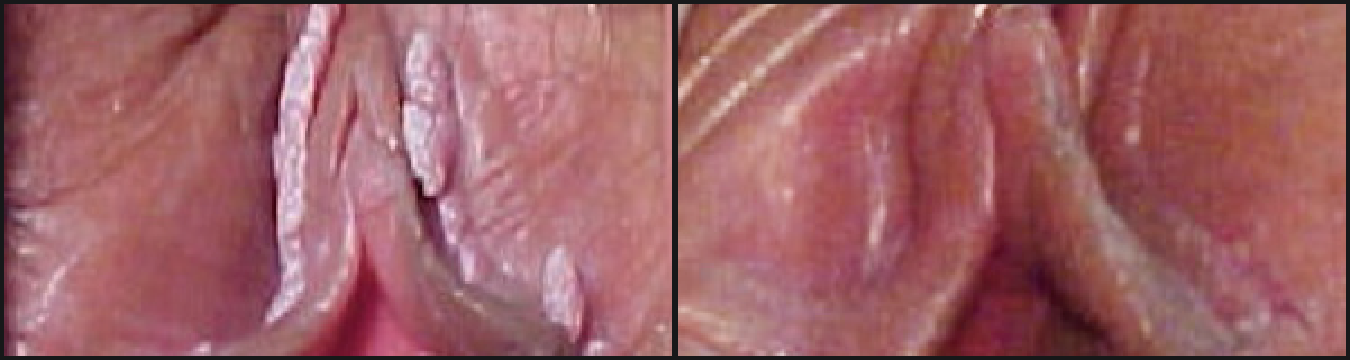
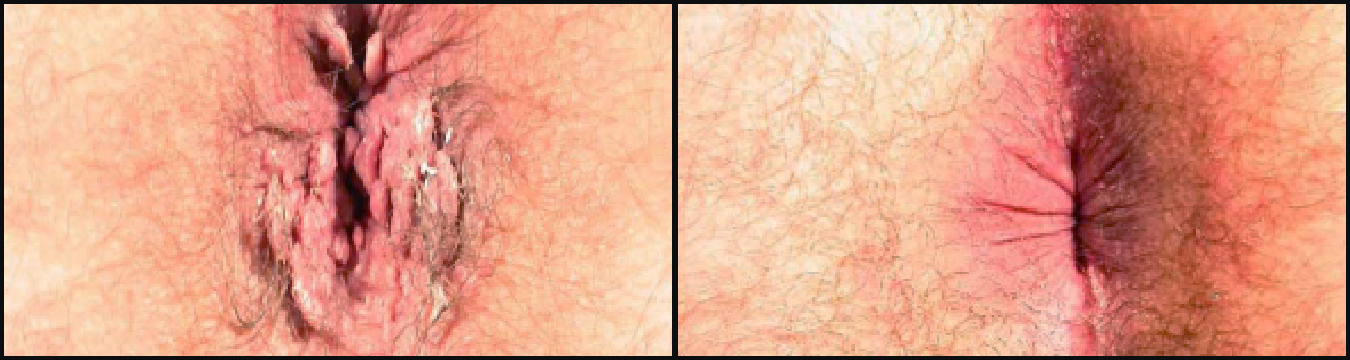
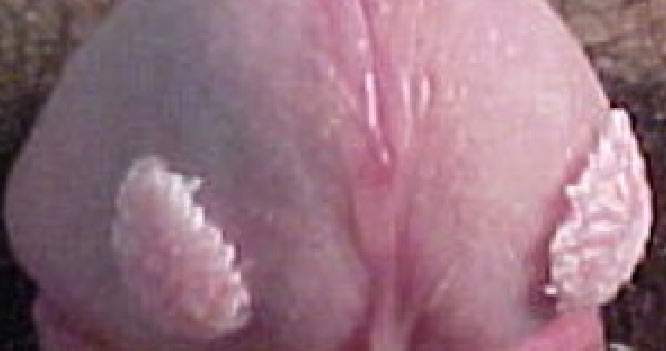
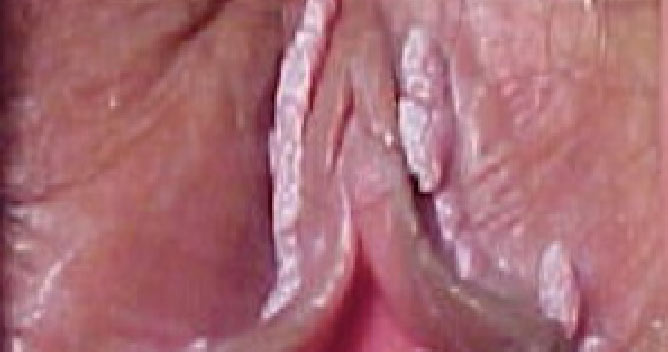
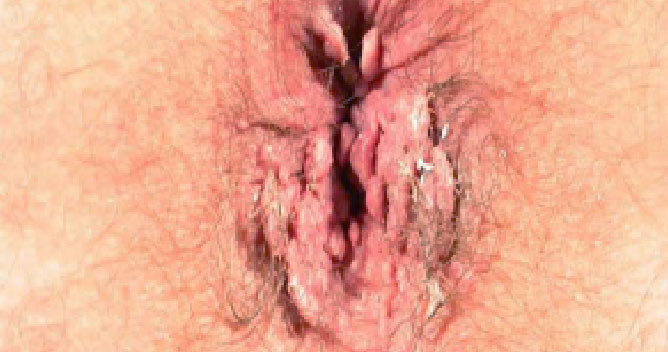
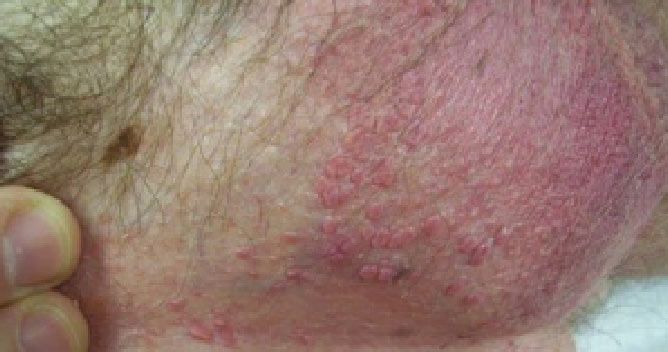
VEREGEN® is indicated for the topical treatment of external genital and perianal warts (Condylomata acuminata) in immunocompetent patients 18 years and older.